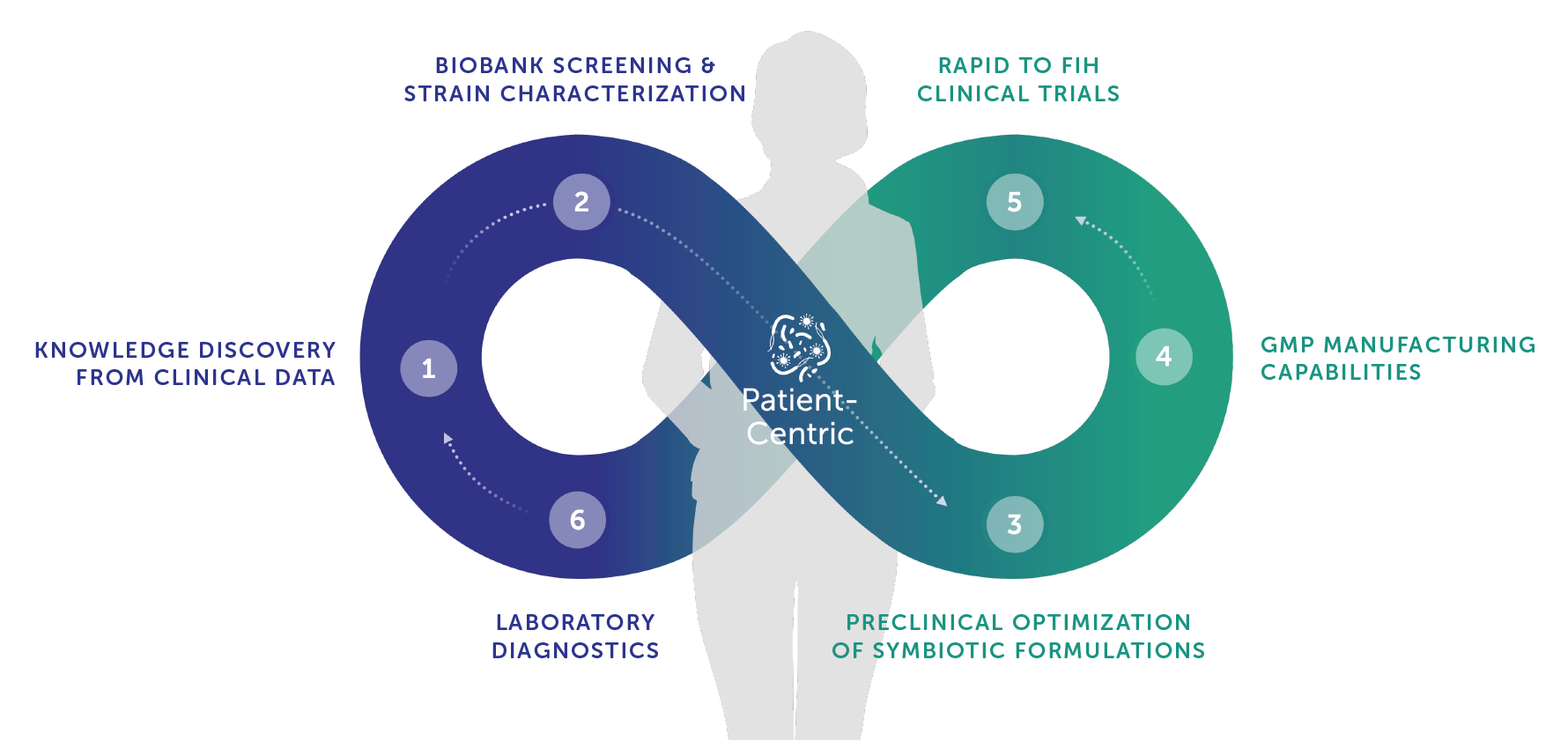
Siolta's Precision Symbiotics Platform Enables Rapid Clinical Evaluation of Lead Consortia
- 1. Knowledge Discovery from Clinical Data

Machine learning and network analysis are applied to high-resolution clinical microbiome data to identify and prioritize therapeutic candidates. Clinical data generated from internal development and external observational studies are collated for increased discovery power.
- 2. Biobank Screening & Strain Characterization
An extensive proprietary biobank derived from healthy donors enables the rapid development of novel biotherapeutics across a wide range of indications. The choice of active ingredient candidates is strengthened by Siolta’s expertise in anaerobic microbiology and microbial genomics.
- 3. Preclinical Optimization of Symbiotic Formulations

In-house translational studies support the evaluation of synergistic functionality of consortia and characterization of disease-specific Mechanism of Action (MoA). Combined in vitro and in vivo studies drive final consortia compositions.
- 4. GMP Manufacturing Capabilities

Internal manufacturing capacity and expertise catalyze efficient advancement of programs into the clinic. Close established partnerships with contract manufacturers translate proprietary tech to scaled production for late-stage clinical and commercial supply.
- 5. Rapid to FIH Clinical Trials > Pharm Market

Rapid advancement into first-in-human studies validates proof-of-concept (e.g., PK/PD & MOA) and de-risks larger late-stage clinical studies. Longitudinal collection of diverse clinical biospecimens improves understanding of immune, metabolic, and microbiome responses.
- 6. Laboratory Diagnostics

Clinical data is leveraged in the development of disease-specific molecular diagnostic tools to improve patient stratification and inform subsequent biotherapeutics development. This approach creates additional value from clinical data generated directly through the LBP development pipeline.
A platform focused on translating microbiome science into novel therapeutics for a healthier world
Siolta’s patient-centric platform leverages our internal expertise in microbiome science, artificial intelligence, anaerobic microbiology, and translational studies to rationally design and optimize live biotherapeutics for the prevention and treatment of diseases with high unmet medical need. Top therapeutic candidates are manufactured in-house using cGMP to achieve pharmaceutical-grade purity, potency, and consistency. Siolta’s streamlined internal capabilities and expertise rapidly drives lead candidates from discovery to proof-of-concept clinical studies. Productive regulatory dialogue and comprehensive nonclinical safety package development allows Siolta to clinically evaluate promising new live biotherapeutic candidates more efficiently, cutting years of development timelines when compared to traditional small molecule or biologic drugs.
A platform with endless opportunities.
We will use our Precision Symbiotics Platform to develop novel live biotherapeutic products for the prevention and treatment of diseases of high unmet medical need.
See Our Pipeline