Product Pipeline
Our unique platform is designed to address multiple targets across a wide range of diseases. With clinical studies underway for allergic diseases, we are broadening our portfolio in the “maternal-infant axis” by advancing programs for recurrent bacterial vaginosis and necrotizing enterocolitis. Siolta’s expanding IP portfolio consists of 9 issued and 29 pending patents.
- STMC-103H (Allergic Diseases)
- STMC-105 (Recurrent BV)
- STMC-106 (NEC)
The Power to Prevent
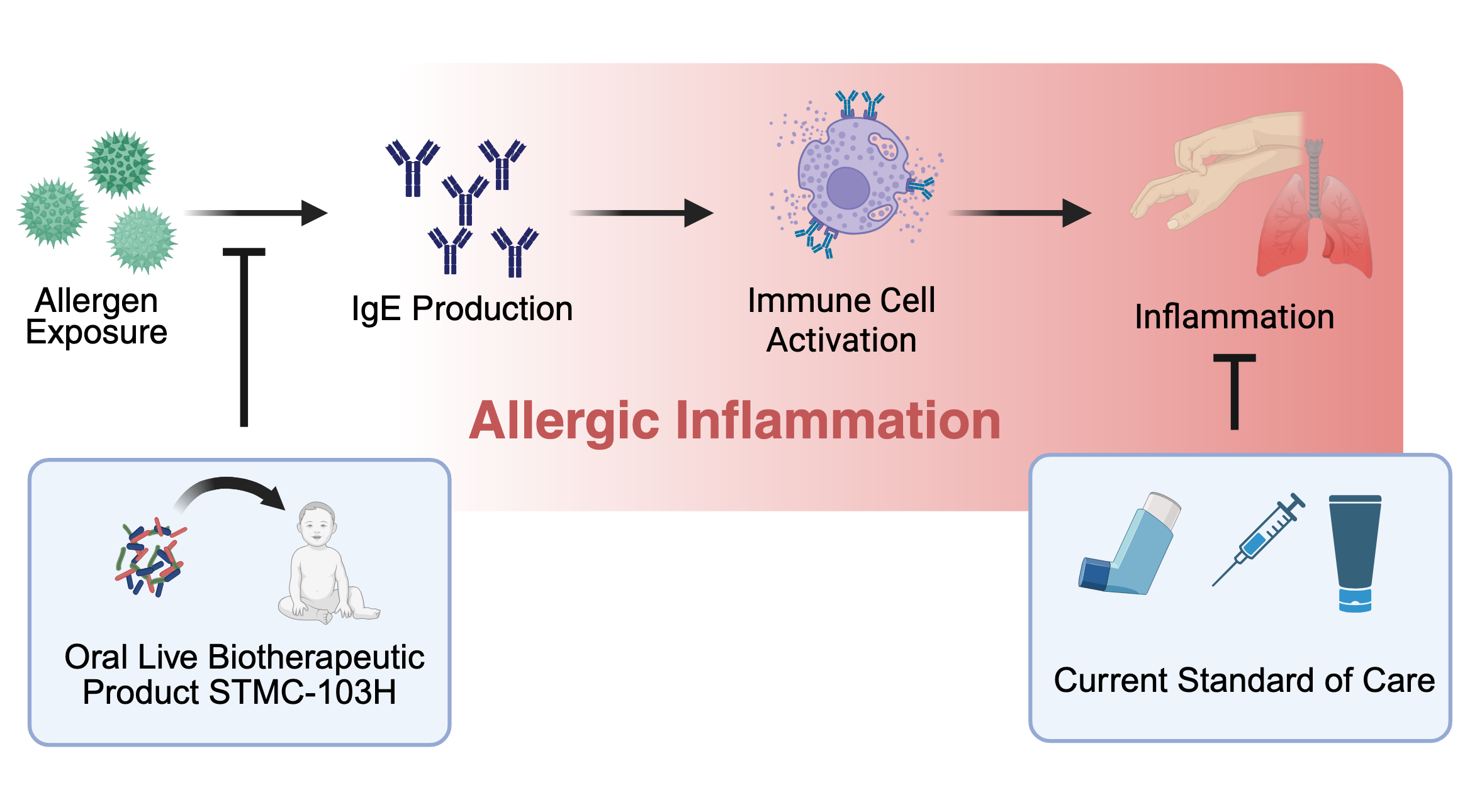
Allergic diseases, including atopic dermatitis (eczema), food allergies, and allergic asthma, arise from immune system dysfunction that triggers the production of IgE antibodies and inflammatory responses to harmless environmental and food substances. These interconnected conditions often develop sequentially in early childhood, a phenomenon known as the “Atopic March.” Siolta Therapeutics has developed STMC-103H, a live biotherapeutic containing a carefully selected mix of beneficial bacteria designed to correct the pro-allergic immune development that occurs in too many infants in our modern world. The potential of STMC-103H to prevent the onset of allergic diseases in newborns and infants is currently being assessed in the groundbreaking Phase 2 ADORED clinical study. This proof-of-concept trial aims to intervene early and prevent allergic disease before it begins by specifically targeting infants at high risk for developing these conditions.
The final participant in the ADORED clinical study completed the trial in November 2025. The final database lock for day 672 and complete study readout is expected in Q1 of 2026.
Allergic Disease Prevention
A New Class of Medicines
Current allergy treatments address symptoms after immune dysfunction has already occurred. Siolta Therapeutics’ microbiome-based approach targets the root cause, aiming to prevent the immune cascade that leads to allergies. Our goal: stop allergic diseases before they start.
Siolta Expanded Access Policy
Siolta Therapeutics is dedicated to advancing our microbiome-based clinical development programs to potentially benefit as many patients as possible. To accomplish this objective, Siolta Therapeutics is currently conducting the well-controlled clinical trials necessary to evaluate safety and efficacy data in clinical trial subjects in support of potential regulatory approval.
Siolta Therapeutics does not offer an expanded access program for our investigational products prior to FDA approval. Expanded access, also called compassionate use, allows access to investigational products to patients with serious or immediately life-threatening diseases who do not meet the enrollment criteria for current clinical trials. This policy has been established as outlined in the 21st Century Cures Act. The expanded access policy may be revised based on data from ongoing and future clinical development.
Siolta Therapeutics recognizes that expanded access to potentially beneficial therapies outside of clinical trials may be valuable to patients with serious conditions, particularly in the absence of effective alternatives treatments. However, at this phase of clinical development, and until sufficient safety and efficacy data are collected, Siolta Therapeutics believes the best approach to characterize and understand the risks and benefits of its investigational products is through the controlled setting of clinical trials. We believe that executing our current and future clinical trials with the highest rigor is the optimal path forward to the regulatory approval that will allow us to ultimately reach the greatest number of patients.
Siolta Therapeutics strongly encourages patients to speak with their physicians about possible clinical trial participation. Reference information about our investigational products and the ongoing clinical trials can be found at https://www.sioltatherapeutics.com/ and https://clinicaltrials.gov. If you have additional questions please speak to your physician or contact us at info@sioltatherapeutics.com.



